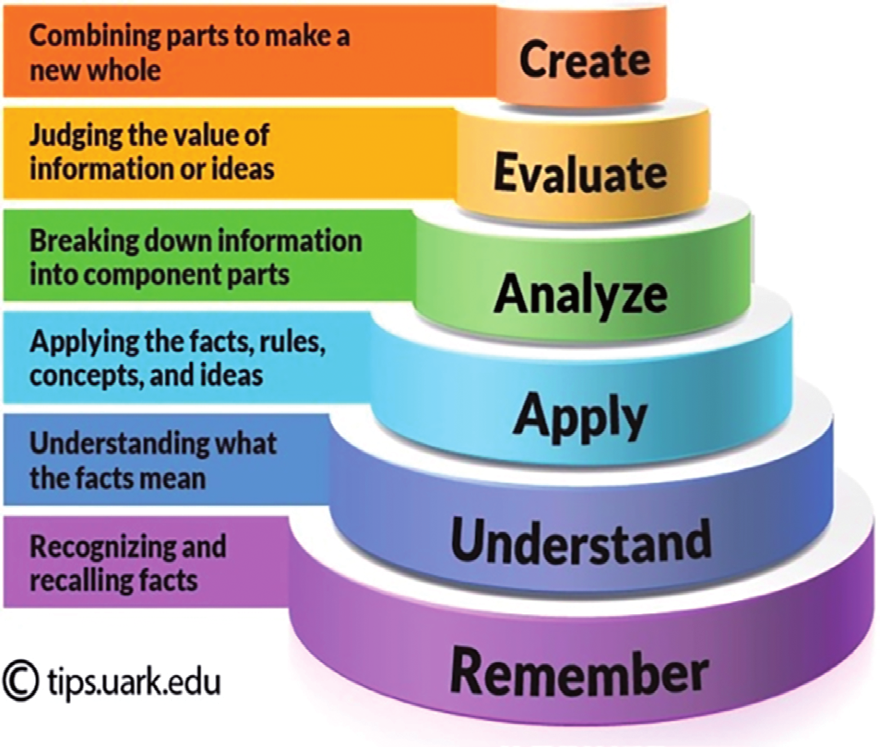
| create | Make a conclusion about the reason for the increase in the entropy of gas with increasing temperature. How does this affect the internal energy of the gas? |
| evaluate | Suggest the condition under which the element carbon (diamond) would have an entropy value of zero. |
| analyse | Compare the 2nd and 3rd electron affinities. Why are they always endothermic? |
| apply | Write equations, including state symbols, that represent the enthalpy change of atomization of chlorine. |
| understand | Explain why the dissolving of silver fluoride in water is always a spontaneous process. |
| remember | Write the equation that shows the relationship between ΔG, ΔH and ΔS for a reaction. |