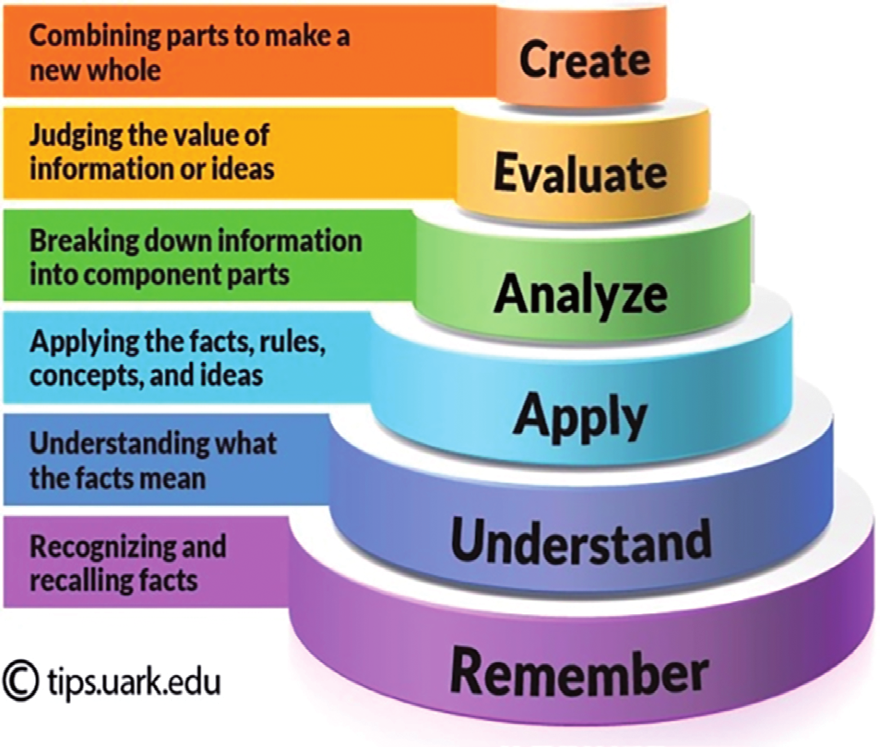
| create | Draw a conclusion about the change in the reactivity of the elements of group II to water and give two reasons for your answer. |
| evaluate | Evaluate and compare the rate of reactions between calcium +water and barium +water. Explain your conclusion. |
| analyse | Both alkaline earth metals and alkali metals tend to _______ electrons to form an ion with a ________ charge. |
| apply | Write the reactions of calcium, calcium oxide with water. |
| understand | Why is it uncommon to find alkaline earth metals in their pure form? |
| remember | As alkaline earth metals have relatively low ionization energies for their first two electrons, what charge do they typically have? |