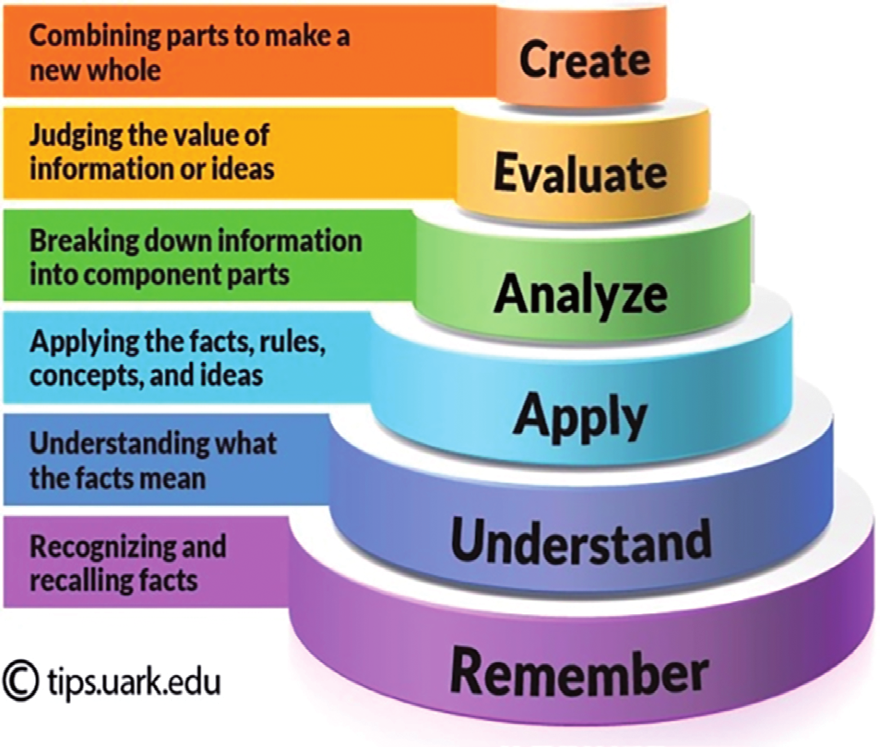
| create | Give three reasons why T.M.S. is a good standard in proton NMR spectroscopy. |
| evaluate | Suggest how butanal and butanone could be distinguished from their carbon-13 nmr spectra. |
| analyse | Suggest how hexane-2-one and hexan-3-one could be distinguished in a mass spectrum. Write equations to show the formation of any important fragment ions. |
| apply | An unknown hydrocarbon has peaks at m/z = 29 and 43, and an M+ peak at m/z = 58 in the mass spectrum. What is the chemical formula for the hydrocarbon? |
| understand | A molecule has no peaks near 3000 cm-1 in its IR spectrum. What can we say about this molecule? |
| remember | What is analytical chemistry? |