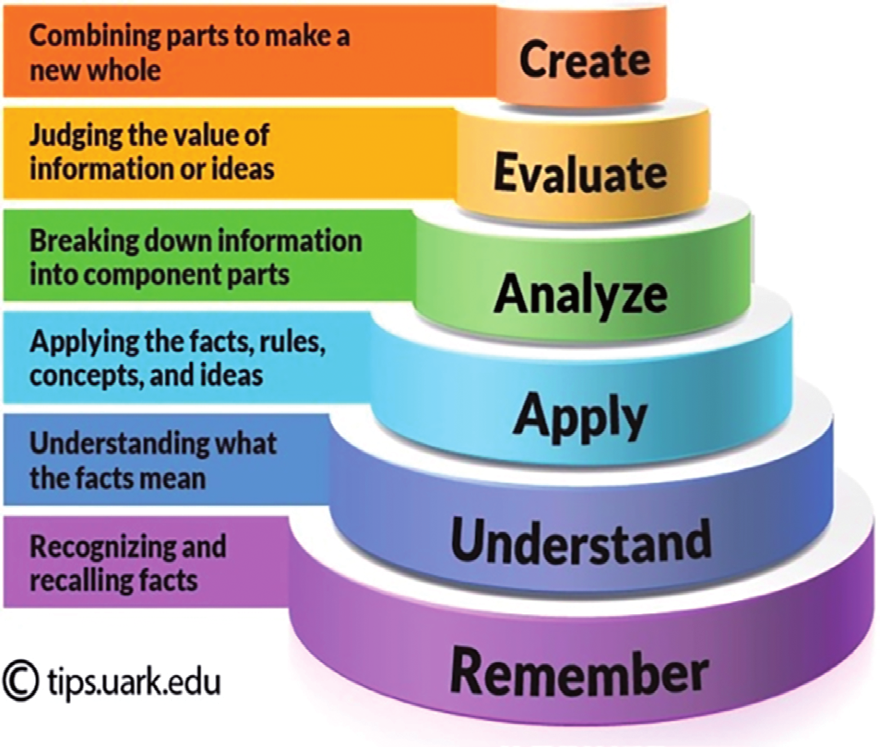
| create | Assess which metals will give stable ionic compounds with halogens. |
| evaluate | Suggest some other properties the halogens might have compared to metals. Eg. strength of solid, heat and electrical conduction? |
| analyse | Which of the halogens is the most reactive? Give an explanation. |
| apply | How many atoms of iodine are needed to react with one atom of magnesium? |
| understand | Explain the group trend at the melting and boiling points of halogens in the group with an increase in the atomic number? |
| remember | What sort of compounds do halogens form when combined with metals? |